Appendix B to Part 425 - Modified Monier-Williams Method
40:32.0.1.1.1.9.2.8.2 : Appendix B
Appendix B to Part 425 - Modified Monier-Williams Method Outline of
Method
Hydrogen sulfide is liberated from an acidified sample by
distillation and purging with nitrogen gas (N2). Sulfur dioxide
interference is removed by scrubbing the nitrogen gas stream in a
pH 7 buffer solution. The sulfide gas is collected by passage
through an alkaline hydrogen peroxide scrubbing solution in which
it is oxidized to sulfate. Sulfate concentration in the scrubbing
solution is determined by either EPA gravimetric test procedure
375.3 or EPA turbidimetric test procedure 375.4 (see 40 CFR 136.3,
Table IB, parameter 65 (49 FR 43234, October 26, 1984, and
correction notice at 50 FR 690, January 4, 1985)).
Apparatus*
(See Figure 1.) * Catalogue numbers are given only to provide a
more complete description of the equipment necessary, and do not
constitute a manufacturer or vendor endorsement.
Heating mantel and control (VWR Cat. No. 33752-464)
1000 ml. distilling flask with three 24/40 joints (VWR Cat. No.
29280-215)
Friedricks condenser with two 24/40 joints (VWR Cat. No.
23161-009)
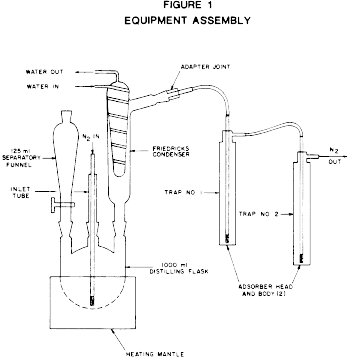
125 ml.
separatory funnel with 24/40 joint (VWR Cat. No. 30357-102) Inlet
tube with 24/40 joint (VWR Cat. No. 33057-105) Adapter joint 24/40
to 19/38 (VWR Cat. No. 62905-26) Adsorber head (2 required) (Thomas
Cat. No. 9849-R29) Adsorber body (2 required) (Thomas Cat. No.
9849-R32) Laboratory vacuum pump or water aspirator Reagents
1. Potassium hydroxide, 6N: Dissolve 340 g. of analytical
reagent grade KOH in 1 liter distilled water.
2. Sodium hydroxide, 6N: Dissolve 240 g. of analytical reagent
grade NaOH in 1 liter distilled water.
3. Sodium hydroxide, 0.03N: Dilute 5.0 ml. of 6N NaOH to 1 liter
with distilled water.
4. Hydrochloric acid, 6N: Dilute 500 ml. of concentrated HCl to
1 liter with distilled water.
5. Potassium phosphate stock buffer, 0.5M: Dissolve 70 g. of
monobasic potassium phosphate in approximately 800 ml. distilled
water. Adjust pH to 7.0 ±0.1 with 6N potassium hydroxide and dilute
to 1 liter with distilled water. Stock solution in stable for
several months at 4 °C.
6. Potassium phosphate buffer, 0.05M: Dilute 1 volume of 0.5M
potassium phosphate stock buffer with 9 volumes of distilled water.
Solution is stable for one month at 4 °C.
7. Alkaline 3% hydrogen peroxide: Dilute 1 volume of 30 percent
hydrogen peroxide with 9 volumes of 0.03N NaOH. Prepare this
solution fresh each day of use.
8. Preparation of stock sulfide standard, 1000 ppm.: Dissolve
2.4 g. reagent grade sodium sulfide in 1 liter of distilled water.
Store in a tightly stoppered container. Diluted working standards
must be prepared fresh daily and their concentrations determined by
EPA test procedure 376.1 immediately prior to use (see 40 CFR
136.3, Table IB, parameter 66 (49 FR 43234, October 26, 1984, and
correction notice at 50 FR 690, January 4, 1985)).
Sample Preservation and Storage
Preserve unfiltered wastewater samples immediately after
collection by adjustment to pH>9 with 6N NaOH and addition of 2
ml. of 2N zinc acetate per liter. This amount of zinc acetate is
adequate to preserve 64 mg./l. sulfide under ideal conditions.
Sample containers must be covered tightly and stored at 4 °C until
analysis. Samples must be analyzed within seven days of collection.
If these procedures cannot be achieved, it is the laboratory's
responsibility to institute quality control procedures that will
provide documentation of sample integrity.
Procedure (See Figure 1 for apparatus layout.)
1. Place 50 ml. of 0.05M pH 7.0 potassium phosphate buffer in
Trap No. 1.
2. Place 50 ml. of alkaline 3 percent hydrogen peroxide in Trap
No. 2.
3. Sample introduction and N2 prepurge: Gently mix sample to be
analyzed to resuspend settled material, taking care not to aerate
the sample. Transfer 400 ml. of sample, or a suitable portion
containing not more than 20 mg. sulfide diluted to 400 ml. with
distilled water, to the distillation flask. Adjust the N2 flow so
that the impingers are frothing vigorously, but not overflowing.
Vaccum may be applied at the outlet of Trap No. 2 to assist in
smooth purging. The N2 inlet tube of the distillation flask must be
submerged deeply in the sample to ensure efficient agitation. Purge
the sample for 30 minutes without applying heat. Test the apparatus
for leaks during the prepurge cycle (Snoop or soap water
solution).
4. Volatilization of H2 S: Interrupt the N2 flow (and vacuum)
and introduce 100 ml. of 6N HCl to the sample using the separatory
funnel. Immediately resume the gas flow (and vacuum). Apply maximum
heat with the heating mantle until the sample begins to boil, then
reduce heat and maintain gentle boiling and N2 flow for 30 minutes.
Terminate the distillation cycle by turning off the heating mantle
and maintaining N2 flow through the system for 5 to 10 minutes.
Then turn off the N2 flow (and release vacuum) and
cautiously vent the system by placing 50 to 100 ml. of
distilled water in the separatory funnel and opening the stopcock
carefully. When the bubbling stops and the system is equalized to
atmospheric pressure, remove the separatory funnel. Extreme care
must be exercised in terminating the distillation cycle to avoid
flash-over, draw-back, or violent steam release.
5. Analysis: Analyze the contents of Trap No. 2 for sulfate
according to either EPA gravimetric test procedure 375.3 or EPA
turbidimetric test procedure 375.4 (see 40 CFR 136.3, Table IB,
parameter 65 (49 FR 43234, October 26, 1984, and correction notice
at 50 FR 690, January 4, 1985)). Use the result to calculate mg./l.
of sulfide in wastewater sample.
Calculations and Reporting of Results
1. Gravimetric procedure:
2. Turbidimetric procedure:
where A = mg./l. of sulfate in Trap No. 2 B =
liquid volume in liters in Trap No. 2 and C = volume in ml. of
waste sample distilled
3. Report results to two significant figures.
Quality Control
1. Each laboratory that uses this method is required to operate
a formal quality control program. The minimum requirements of this
program consist of an initial demonstration of laboratory
capability and the analysis of replicate and spiked samples as a
continuing check on performance. The laboratory is required to
maintain performance records to define the quality of data that is
generated. Ongoing performance checks must be compared with
established performance criteria to determine if the results of
analyses are within precision and accuracy limits expected of the
method.
2. Before performing any analyses, the analyst must demonstrate
the ability to generate acceptable accuracy and precision by
performing the following operations.
(a) Perform four replicate analyses of a 20 mg./l. sulfide
standard prepared in distilled water (see paragraph 8 under
“Reagents” above).
(b)(1) Calculate clean water precision and accuracy in
accordance with standard statistical procedures. Clean water
acceptance limits are presented in paragraph 2(b)(2) below. These
criteria must be met or exceeded before sample analyses can be
initiated. A clean water standard must be analyzed with each sample
set and the established criteria met for the analyses to be
considered under control.
(2) Clean water precision and accuracy acceptance limits: For
distilled water samples containing from 5 mg./l. to 50 mg./l.
sulfide, the mean concentration from four replicate analyses must
be within the range of 72 to 114 percent of the true value.
3. The Method Detection Limit (MDL) should be determined
periodically by each participating laboratory in accordance with
the procedures specified in “Methods for Chemical Analysis of
Municipal and Industrial Wastewater,” EPA-600/4-82-057, July 1982,
EMSL, Cincinnati, OH 45268. For the convenience of the user, these
procedures are contained in appendix C to part 425.
4. A minimum of one spiked and one duplicate sample must be run
for each analytical event, or five percent spikes and five percent
duplicates when the number of samples per event exceeds twenty.
Spike levels are to be at the MDL (see paragraph 3 above for MDL
samples) and at x when x is the concentration found if in excess of
the MDL. Spike recovery must be 60 to 120 percent for the analysis
of a particular matrix type to be considered valid.
5. Report all results in mg./liter. When duplicate and spiked
samples are analyzed, report all data with the sample results.
[53 FR 9184, Mar. 21, 1988]